HISTOPATHOLOGICAL EVALUATION OF PARANASAL SINUS OPACITIES ON TEMPORAL BONE TOMOGRAPHY IN PATIENTS WITH CHRONIC OTITIS MEDIA
2Yıldırım Beyazıd Üniversitesi Tıp Fakültesi, KBB, Ankara, Turkey
Summary
Objective: This study is designed to analyse asymptomatic sinus opacities histopathologically in comparison to chronic rhinosinusitis(CRS).Material and Method: This is a prospective study conducted in the Department of Otolaryngology between December2008- November 2009. From 20 patients who have asymptomatic paranasal sinus opacities on temporal computed tomography (CT), who were selected during their preoperative evaluation out of 147 chronic otitis media (COM) patients, maxillary sinus biopsies were taken intraoperatively from asymptomatic paranasal sinus opacities after timpanoplasty/ timpanomastoidectomy. Control grup consisted of biopsies of 8 patients who have functional endoscopic sinus surgery (FESS) for CRS. Histopathologic analysis and comparison were made statistically between two grups.
Results: Lenfoid follicule formation, congestion and pathological grade were found to be different between the groups (p=0,017, p=0,011, p=0,013).Epithelial integrity (p=0.072), goblet cell hyperplasia(GCH) (p=0.238), squamous metaplasia (p=0.320), thickness of basement membrane (BM) (p=0.11), submucosal gland hyperplasia(SMGH) (p=0.089), eosinophils (p= 0.328), neutrophils (p=0.381), lymphocytes (p=0.438), plasma cells (p=1.00), macrophages (p=0.55), calcification (p=0.063), lymphatic dilatation (p= 1.00), subepithelial edema (p= 0.285), polipoid degeneraion (p=1.00) were statistically similar in both groups. Similarly, the calcification detected by pathology and CT were not significantly correlated (κ=0.379 ve p=0.075).
Conclusion: Asymptomatic incidental paranasal sinus opacities seen on temporal CT in patients of chronic otitis media are similiar to CRS histopathologically.
Introduction
Diagnosis of CRS is mostly based on subjective symptoms. According to the guidelines of American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) in 2007; diagnosis of CRS is based on symptom criterias and objective findings[1,2]. CRS is defined as 2 or more of major symptom criteria or 1 major criteria and 2 or more minor criterias for a period of 12 weeks or longer. Additionally, endoscopic findings such as nasal polyps, purulent nasal discharge in the middle meatus or ethmoid region or radiographic diagnosis of CRS is also adviced. In the presence of these criterias, asymptomatic paranasal sinus opacities cause diagnostic dilemma[3,4].Clinical symptoms of sinusitis do not always correlate with CT findings. However, there is not any problem when symptomatic patients with positive sinus CT and asymptomatic patients with negative sinus CT are scanned [5,6]. The problem occurs with the symptomatic patients with negative CT and asymptomatic patients with positive CT. According to some researchers, this discrepancy is attributed to the high sensitivity and moderate specificity of CT [7,8]. 30 % of asymptomatic patients show incidental mucosal changes on CT in adults. Furthermore, this ratio is higher in children [9]. Are asymptomatic sinus opacities inflamation and do they affect chronic otitis media development and maintenance? In this study, we take biopsies from maxillary sinus lesions of patients who do not have symptoms of nasal and sinus infections, and in whom incidental opacities at maxillary sinuses were detected on temporal CT. We aimed to investigate whether asymptomatic or silent sinus infection real infection and accompanies COM.
Methods
This study was prospectively conducted in accordance with the ethical principles stated in the Declaration of Helsinki and was approved by the Ethics Committee for Clinical Research of the Ankara Atatürk Education and Research Hospital (No:2009/05/08). All participants included in the study in our case group, were between the ages of 13 and 61 undergoing timpanoplasty/ tympanomastoidectomy. They were selected from 147 COM patients during their preoperative evaluation in Ear Nose and Throat Clinic-1 of Ankara Atatürk Education and Research Hospital. These 20 patients did not have symptoms of sinusitis, without any score in SNOT-20, but mucosal thickening was incidentally detected at their paranasal sinus on temporal CT during their pre-operative evaluation. Two separate informed consent; one for tympanomastoidectomy, the other for maxillary sinus biopsy, were obtained from each volunteer or legal guardian of each participant following a detailed explanation of the study protocol and the risks and contributions of their participation in this study. Exclusion criteria were symptomatic sinusitis, allergic rhinitis, asthma, mucociliary disorders like Kartagener syndrome and immotile cilia. The control group consisted of microscopic slides of 8 patients who have FESS for CRS. The patients were identified retrospectively by chart review.Pre-operative temporal CT scans were made during patient evaluation during clinical examination for chronic otitis. All scans were made patients lying on their backs with the arms at the sides without using contrast. Scans on temporal CT was performed with a 64 slide multi-detector CT scanner (Toshiba Aquilion, Chicago, USA). Imaging properties are as follows: The tube potency used was 120 kv and the tube current was 50 mAS. The slice thickness is 1 mm and in bone window at sagittal scans making coronal and sagittal multiplanar reconstruction. All paranasal sinuses are included in views of scans and CT scans are interpreted under guidance of Radiology Department for the study for proper patient selection.
Under general anesthesia, as soon as completion of timpanoplasty/ timpanomastoidectomy, maxillary sinus biopsies were taken from these 20 selected asemptomatic sinusitis patients, intraoperatively. For all 20 patients in study group, mucosal thickenings on paranasal sinuses were obtained and collected. The biopsies were taken through a puncture made on fossa canina by trocar for entrance to maxillary sinus.After inspection with 0 degree endoscope, the biopsies were taken from maxillary sinus from silent paranasal sinus mucosal thickenings.
Under guidance of Pathology Department, pathology specimens taken from our case group of 20 patients were fixed in %10 formaldehyde, followed by dehydration in graded alcohol solutions. Thereafter, they were embedded in paraffin blocks, serially cut into 5µm thick sections, and put on glass slides and stained with H&E. H&E stained sections were examined with a light microscope under x400 magnification to count inflammatory cells. In the case group and the control groups, 17 parameters are evaluated microscopically. The histopathological parameters are epithelial integrity, GCH (Fig 1), squamous metaplasia, thickness of BM, SMGH (Fig 2), eosinophils (Fig 3), plasma cells, neutrophils, lymphocytes, macrophages, lenfoid follicle formation, lymphatic dilatation, congestion, subepithelial edema, calcification (Fig 4), and polypoid degeneration. Pathological grading is the last parameter to be evaluated. Mucosal biopsy specimens were evaluated at the sides, which has most inflammated by each parameter. Also, each parameter were also assessed in control group, slides of FESS patients at x400 magnification under microscope. Histopatological grading was used in GCH, thickness of BM, eosinophils, neutrophils, lymphocytes, SMGH, pathological grade (Table 1). Study and control groups, 17 parameters were statistically analyzed. Histopathological calcification and radiologic calcification are also compaired.
 Büyütmek İçin Tıklayın |
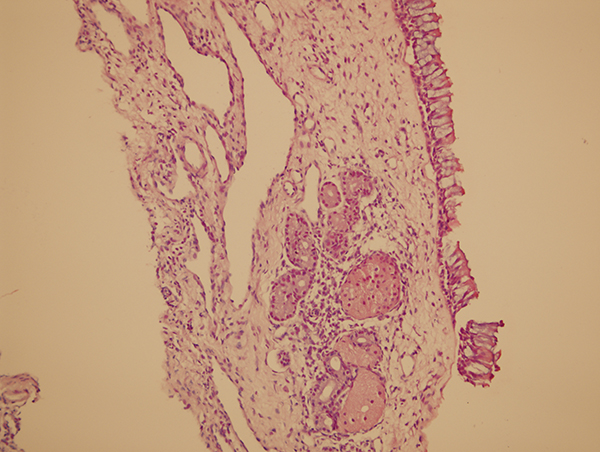
Fig 1: Goblet cell hyperplasia, lymphatic dilatation, lymphocytes, submucosal glands, oncocytic changes are seen in mucosal biopsies from asymptomatic paranasal sinus opacities. H&E, x 100. |
 Büyütmek İçin Tıklayın |
Fig 2: Submucosal gland hypertrophy and goblet cell hyperplasia H&E , X40. |
 Büyütmek İçin Tıklayın |
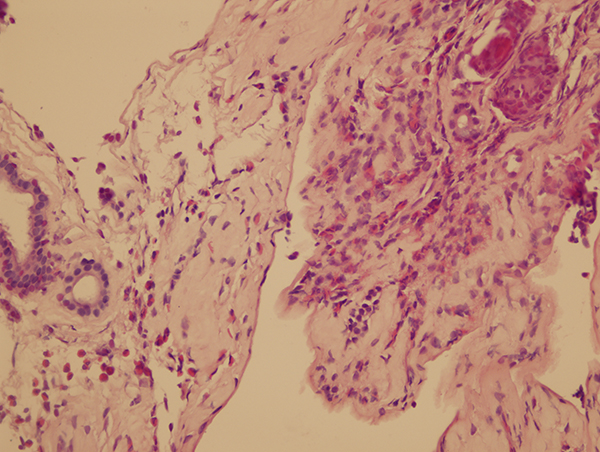
Fig 3: On submucosal region, eosinophilic infiltration . H&E, x400. |
 Büyütmek İçin Tıklayın |
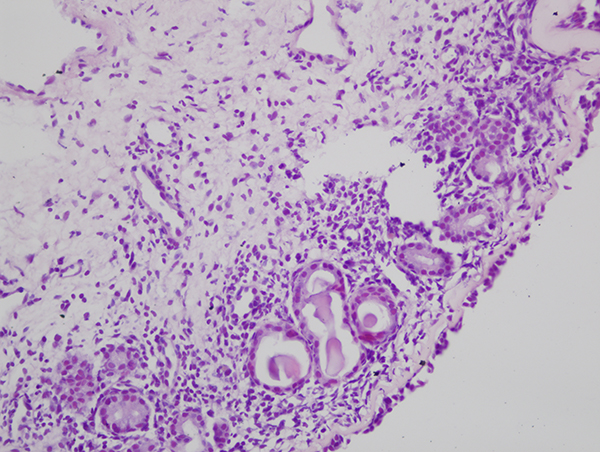
Fig 4: Dilatation of submucosal glands, calcification in the glands, perivascular lymphocytes, BM thickening. H&E, x 100. |
Data analysis was performed with SPSS for Windows 11.5 package program. Descriptive statistics were expressed as mean and ± standard deviation for continuous variables, and nominal and ordinal variables were expressed as number of cases and percent (%). The significance of the difference between the groups in terms of mean age was evaluated with Student's t-test. Ordinal variables were examined by Mann-Whitney U test. Nominal variables were evaluated by Pearson's Chi-Square test or Fisher's exact test. The results were considered statistically significant for p <0.05.
Results
The study group consists of 20 patients. 9 of them (%45) are female and 11 (%55) of them are male. Their ages vary between 13-61 years old. The mean age was 37±16. The control group consists of 8 patients, 4 of whom are female, and 4 of whom are male. Their ages vary between 19-61 years old. Their mean age was 35,7± 14,7. There were no statistical significance between the groups in terms of the age and the gender ( p=0.844, p=1,000).There were no statistical significance in epithelial integrity, GCH, squamous hyperplasia, thickness of BM, SMGH, eosinophil, neutrophil, lymphocyte, plasma cells, macrophage, calcification, lymphatic dilatation, subepithelial edema, polypoid degeneration between the groups (p<0.005). Lenfoid follicle formation, the congestion, pathologic grade were significantly different (p=0.017, p=0.011, p=0.013) (Table 2,3,4) Calcification detected by pathology and CT were not significantly correlated (κ=0.379 and p=0.075).
Tablo 3: Statistical analysis and distribution of lymphoid cells in case and control group.
Discussion
As well as rinosinusitis and the otitis media with effusion (OME) have common features, we see their coexistance in the literature. Paranasal sinuses, eustachian tube and in some parts of middle ear cavity are lined by ciliated pseudostratified columnar epithelium. This epithelium is responsible for mucociliary Clearance (MCC) which is a defence mechanism clearing inhaled particles and pathogens from the unified airway. The middle ear and paranasal sinus submucosa both have goblet cells and mucus producing cells[10,11]. The paranasal sinuses drainage is by maxillary ostium and middle ear ventilation and drainage is by eustachian tube. Obstruction of these openings leads to infection of the cavity behind them. Same flora such as Haemophilus influenzae, Streptococcus pneumonia, prevotella species, peptostreptococcus species cause inflammation of middle ear cavity and paranasal sinuses. Treatment of one disease can cure the other[12]. Both diseases are caused by bacterial infection, allergic and immunologic factors, adenoid hyperplasia and upper airway infections[13]. In Hoshaw and Nickman's study, 43% of 166 children with otitis media necessitating ventilation tubes had radiographic maxillary sinusitis. Grote and Kuypers found that %45 of 1252 children with chronic OME had maxillary sinusitis and %82 of children with chronic OME get rid of disease by treatment of sinusitis[14]. Finkelstein et all investigated 148 patients with CRS and OME, 29 of them both have CRS and OME. On endoscopic examination of 29 patients mucopurulent discharge is seen and they reported that OME is a presenting symptom of chronic sinusitis, not a distinct clinical entity[12,14]. On the contrary, there are also studies against coexistence of CRS and OME. Kim et all in a national wide study performed in Korea found no relation between CRS and COM[15]. The association of CRS and OME or COM is not yet clearly identified.Eustachian tube is a fibrocartilaginous tube which has many functions, the physiology of which is still not fully known. Optimization of middle ear sound transmission and protection of the inner ear structures pressure regulation within middle ear cleft are some of its functions[16]. Mechanical protection from rhinofarengial secretions and pathogenic microorganisms, local immune defence and mucociliary clearence (MCC) are also function of eustachian tube. MCC is affected by systemic and local disorders. One example of systemic disorders affecting MCC is PCD. CRS, adenoid hypertrophy are local factors contributing to MCC disorder. In PCD, OME is very common and mild- moderate conductive hearing loss is observed[11]. Otologic complications like COM, retraction pockets, cholestatoma and tympanic membrane perforation were also seen[17]. MCC is one of the functions of tubotympanic system which is coordinated by internal pharyngeal part of eustachian tube and lower part of its lumen[18]. Ars et al stated that in middle ear posterosuperior,attic and mastoid, mucosa is lined by flat endothelial cells with vessels near the basal membrane in which gas exchange is carried out. In anteroinferior part of middle ear, which is lined by multistratified ciliary epithelium and take part in MCC. In addition to MCC, tubotympanic system,eustachian tube and mastoid cavity, have pressure regulation function by the tympanic membrane receptors for pressured eustachian tube and its peritubal musculature,and also have gas exchange, protective properties and immune defence functions[19] Tubal function is an intrinsic and important component of the middle ear cleft pressure regulation system The tubal muscles must actively dilate the tubal valve open in order for adequate gas exchanges into the middle ear cleft to occur. Tubal dilation is likely naturally facilitated by the presence of surface tension? reducing substances that are found in its mucus. Surfactants are produced within the tubal mucosa and probably aid in reducing the surface tension of the lumen, which reduces the work required to dilate the tube[16,20]. In 2010, in cadaver experiences feasibility of Eustachian tuboplasty baloon tuboplasty(BDET) was shown by Ockermann and his colleques. Over last 10 years, dynamic fuction of eustachian tube was experienced by BDET[21]. Nevertheless, the unified airway system containing eustachian tube and tympanomastoid cavities functions are not fully explained and the unexplained secrets within the unified system will lighten the dilemmas and unknowns in our silent sinus infections and its relation to tympanomastoid effects.
Microscopic findings of chronic sinusitis are submucosal edema, mixed inflammatory infiltrate consisting of lymphocytes, plasma cells, eosinophils, histiocytes and occasionally neutrophils. In longstanding cases surface mucosa shows squamous metaplasia, goblet cell hyperplasia,subepithelial edema,submucous gland formation, mucosal thickening, papillary hyperplasia of the mucosa, BM thickening[22,23,24,25]. In histopathological study of Anselmo-Lima et all, histopatological examination is performed in biopsies taken during FESS operation. Maxillary sinus mucosa show loss of ciliated mucosa with the development of atypical metaplastic epithelium which can cause functional abnormalities within the paranasal sinuses. In submucosa, inflammatory infiltrate of lymphocytes, plasma cells, eosinophils, histiocytes, mast cells were seen producing various cytokines. Subepitelial thickening, basal membrane hyalinization were also seen. In control biopsies with recurrence of disease (group 1) and with the group of patients biopsies taken with remission of disease one year later (group 2). The histopathologic findings persist in group 1, whereas partial recovery were seen group 2 with pseudostratified epithelium, goblet cell hyperplasia, some loss of ciliated cells in electron microscopy (EM)[26]. Chan et all in their histopathological study of young children and adult CRS patients,find same histopathological parameters in both group of their work, but children had higher density of submucosal lymphocytes, lower density of submucosal eosinophils, thinner and more intact epithelium, thinner basement membrane and fewer submucosal mucous glands[27]. In our study, we observe statistically similar findings in mucosal biopsies of incidental opacities and biopsies taken after CRS taken during FESS operation. Our study can be detailed by studying under EM, studies of staining of mediators of infection, autoimmunity and allergy.
According to expert panel formed by the American Academy of Allergy and Immunology (AAAAI), the American Academy of Otolaryngology Allergy (AAOA), AAO-HNS, American College of Allergy, Asthma and Immunology (ACAAI) and the American Rhinologic Society(ARS) in 2004, rinosinusitis is divided into four groups; acute (bacterial) rhinosinusitis, chronic rhinosinusitis without polyps (CRSsNP), CRS with polyps (CRSwNP) and allergic fungal rhinosinusitis(AFRS). The theory under CRS was sinus obstruction and bacterial infection gives its place to appreciation that CRS has inflammatory component that might be triggered simultaneously or independently by some factors such as; biofilms and osteitis, allergy and other disorders of immunity, intrinsic factors of upper airway, superantigen from Staphylococcus aureus in CRSwNP, colonizing fungi that induce and sustain eosinophilic inflammation and metabolic perturbations like aspirin sensitivity[1]. Pathophysiology of CRSsNP, CRSwNP and AFRS is not fully understood and these groups intermingle with each other[28]. Histologic features of nasal polyp tissue are epithelial damage, thickened BM, mostly edematous to fibrotic stroma, reduced number of vessels, glands and no neuronal structures. Early features of NP is presence of eosinophils forming subepithelial cap over a pseudocyst filled with albumin[1]. CRSwNP is the most common form of CRS. Allergic mucin and polyp formation is not seen in it. In CRSwNP there is a strong tendency of recurrence after surgery, eosinophil and IL-5 excess in CRSwNP in the USA and Europe. But it is not the case for Japan and East Asia. In these areas more than half of CRSwNP do not exhibit eosinophils[29]. AFRS is a subgroup of noninvasive fungal rinosinusitis. It is a type I hypersensitivity reaction to extramucosal sinus fungal hypae[30]. In AFRS, major diagnostic criteria by Bent and Kuhn includes type 1 hypersensitivity,nasal polyposis, CT, findings, eosinophilic mucin, positive fungal stain. Minor criterias asthma, unilateral disease,bone erosion, fungal cultures,Charcoat Leyden cristals,serum eosinophilia[31]. Eosinophilic CRS is another topic of classification. Under the name of eosinophilic CRS; super antigen-induced eosinophilic CRS, AFS, nonallergic fungal eosinophilic CRS, and aspirin-exacerbated eosinophilic CRS[32]. In 5 of our biopsies taken from incidental opacities also eosinophils are seen. In control group 2 patients had polyp formation with eosinophilia infiltration.
Calcification in the sinuses or intralesional hyperintensty is seen in especially fungal diseases as well as it is seen in nonfungal diseases. The unilateral opacisification of maxillary sinus with intralesional hyperintensity or calcification is suggestive of maxillary sinus fungus ball in fungal sinusitis. Yvon et all in their study on 471 patients, sinus calcification is observed in 16 (%3) of nonfungal sinusitis, whereas 39(%51) patients had fungal sinusitis. The calcification is seen in the centre of sinus in fungal sinusitis, on the other hand, in nonfungal sinusitis, it is seen at the periphery of sinus cavity[33]. In our study, all of the calcifications take place peripherally which favors nonfungal sinusitis. Fungal spores are ubiquitous in the environment and inhaled with breath in air. Only minority elicits a pathogenic immunologic response and invasive forms of fungal infections are extremely rare. In cases of fungal sinusitis, calcium and magnesium salts become deposited in areas of fungal growth and fungus infected mucin. These collections appear hyperintense on non-contrast CT. Allergic mucin in fungal sinusitis starts an local inflammatory response, erosive changes of sinus wall sinus expansion with bone remodeling.[34,35,36] Biopsies from incidental opacities in our study show %21,1 calcification on CT, whereas microscopic calcification is seen in %36.8. It is worth to mention that we can not see small calcifications on CT.
Silent or subclinical otitis media is an entity which has been described in 1979 by Paparella. It is a chronic pathological condition that is clinically undetectable behind an intact tympanic membrane. The lack of symptoms and therefore lack of treatment leads to risk of complications in silent otitis media. Inflammatory reaction in silent sinusitis cause granulation tissue, tympanosclerosis, cholestrol granuloma, cholestatoma, inclusion cyst, bone necrosis. These changes can heal spontanously or cause intracranial or extracranial manifestations of COM[37]. As in the case of silent otitis media, does subclinical sinusitis in our incidental opacities cause remote effects in temporal bone by having been unexplained immunological, inflammatory mechanisms?
Given that complex, unsolved mechanisms play a role in the unified system of upper airway, otitis must be investigated in sinusitis patients or vice versa. In our study, we took maxillary sinus biopsies intraoperatively from asymptomatic paranasal sinus opacities after timpanoplasty/timpanomastoidectomy from 20 patients who have asymptomatic paranasal sinus opacities on temporal CT, who were selected during their preoperative evaluation. The control group consisted of biopsies of 8 patients who have FESS for CRS. In histopathologic analysis and comparison, two groups were found to be alike. Therefore, we concluded that like silent otitis media there is silent sinusitis. Although, the complex mechanisms under all kinds of CRS are not fully understood. Secrets under polyp development, AFRS, calcification, and all other unexplained inflammatory events give clues for explanation of pathogenesis and treatment of CRS.This asymptomatic sinusitis cause microbiological colonisation of adenoid and impairs MCT in Eustachian tube and affects tubotympanic system and leads to OME and refractory COM and slow down eradication of them. Moreover, it may influence success of tympanoplasty operations. The incidental paranasal sinus opacities can be searched in unsuccesful tympanoplasty operations and local medications like nasal steroids, etc. can be added to enhance success of tympanoplasty/timpanomastoidectomy operations.
Acknowledgements: We would like to thank to our colleagues at Department of Radiology, Pathology and Sherring&Plough for sponsorship.
Funding: This study received grant from Sherring Plough for expences of the study.
Conflict of interest: The authors declares that they have no conflict of interest pertaining to this manuscript.
Reference
1) Meltzer EO, Hamilos DL, Hadley JA et all. Rhinosinusitis: Establishing definitions for clinical research and patient care Otolaryngol Head-Neck Surgery2004; 131:6 suppl; s1-62. [ Özet ]
2) Kenny TJ, Duncavage J, Bracikowski J, Yıldırım A, Murray J, Tanner SB Prospective analysis of sinus symptoms and correlation with paranasal computed tomography scan. Otolaryngol Head Neck Surg 2001; 125(1):40-3. [ Özet ]
3) Rosenfeld RM, Picirillo JF, Chandrasekhar SS, Brook I et all. Adult Practice Guideline (Update): Adult sinusitis. Otolaryngol Head Neck Surg 2015; 152(2S): S1-39. [ Özet ]
4) Fokkens W, Lund VJ, Mullol J et all EPOS 2012: European position paper on rhinosinusitis and nasal polyps 2012. A summary for otolaryngologists. Rhinology 2012;50(1):1-12. [ Özet ]
5) Deoshale NV, Khadakkar SP, Harkare VV.et all. Diagnostic accuracy of nasal endoscopy as compared to computed tomography in chronic sinusitis. Indian J. Otolaryngol Head Neck Surg 2017;69(4):494-499. [ Özet ]
6) Bhattacharyya N, Fried MP The Accuracy of Computed Tomography in the Diagnosis of Chronic Rhinosinusitis. Laryngoscope 2003;113(1):125-9. [ Özet ]
7) Bhattacharyya N Radiographic stage fails to predict symptom outcomes after endoscopic sinus surgery for chronic rhinosinusitis. Laryngoscope 2006;116(1):18-22. [ Özet ]
8) Witkopf ML, Beddow PA, Russell PT, Duncavage JA, Becker SS Revisiting the interpretation of positive sinüs CT findings: a radiological and symptom-based review. Otolaryngol Head Neck Surg 2009;140(3):306-11. [ Özet ]
9) Jones NS CT of the paranasal sinuses: a review of the correlation with clinical, surgical and histopathological findings. Clin Otolaryngol Allied Sci 2002;27(1):11-17. [ Özet ]
10) Damar M, Dinç AE, Erdem D, Bişkin S, Eliçora SŞ, Kumbul YÇ The Role of the Nazal and Paranazal Sinus Pathologies on the Development of Chronic Otitis Media and its Subtypes: A computed tomography study. Nigerian J Clin Pract 2017;20(9): 1156-60. [ Özet ]
11) Yazıcı H Nasal mucociliary clearance in adenoid hypertrophy and otitis media with effusion. Curr Allergy Asthma Rep 2015;15(12):74. [ Özet ]
12) Hong CK, Park DC, Kim SW, Cha Cl, Cha SH, Yeo SG Effect of paranasal sinusitis on the development of otitis media with effusion: influence of eustachian tube function and adenoid immunity. Int J Pediatr Otorhinolaryngol 2008;72(11):1609-18. [ Özet ]
13) Quaranta N, Iannuzzi L, Gelardi M Does the type of rhinitis influence development of otitis media with effusion in children? Curr Allergy Asthma Rep 2014;14(11):472. [ Özet ]
14) Hong S, Lee WH, Lee SH, Rhee CS, Lee CH, Kim JW Chronic rhinosinusitis with nasal polyps is associated with chronic otitis media in the elderly. Eur Arch Otorhinolaryngol 2017;274(3):1463-70. [ Özet ]
15) Kim CS, Jung HW, Yoo KY Prevalance and risk factors of chronic otitis media in Korea:results of a nation-wide survey. Acta Otolaryngol 2013;113(3):369-75. [ Özet ]
16) Ars B, Dirckx J Eustachian tube function. Otolaryngol Clin N Am 2016;49(5):1121-1133. [ Özet ]
17) Kreicher KL, Schopper HK, Naik AN, Hatch JL, Meyer TA Hearing loss in children with primary ciliary dyskinesia. Int J Pediatr Otolaryngol 2018;104:161-5. [ Özet ]
18) Takeuchi K, Kitano M, Sakaida H, Usui S, Masuda S, Oqawa S, Ikejiri M, Nagao M, Fujisawa T, Nakatani K Analysis of otolaryngologic features of patients with primary ciliary dyskinesia. Otol Neurotol 2017;38(10):e451-e456. [ Özet ]
19) Martin C, Karkas A, Prades JM Tubotympanic system functioning. Eur Ann Otolaryngol Head Neck Dis 2017;134(3):177-184. [ Özet ]
20) Dean M, Pynnonen MA In-Office balloon dilatation of the Eustachian tube under local anesthesia:A retrospective review.Otolaryngol Clin North Am 2019;52(3):509-520. [ Özet ]
21) Ockermann T, Reineke U, Upile T, Ebmeyer J, Sudhoff HH Balloon Dilatation Eustachian Tuboplasty:A feasibility study Otol Neurotol 2010;31(7):1100-1103. [ Özet ]
22) Montone KT, Livolski V Inflammatory and Infectious Lesions of the Sinonasal Tract. Surg Pathol Clin 2017;10(1):125-154. [ Özet ]
23) Cousin JN, Har-El G, Li J (2000) Is there a Correlation between radiographic and histologic findings in chronic sinusitis? J Otolaryngol 2000;29(3):170-3. [ Özet ]
24) Dhong H, Kim H, Cho D Histopathologic characteristics of chronic sinusitis with bronchial asthma. Acta Otolaryngol 2005;125(2): 169-76. [ Özet ]
25) Soler ZM, Sauer DA, Mace J, Smith TL Relationship between clinical measures and histopathologic findings in chronic rhinosinusitis. Otolaryngol Head and Neck Surg 2009;141(4): 454-61. [ Özet ]
26) Anselmo-Lima WT, Ferreira MD, Valera FC, Rossato M, de Mello VR, Demarco RC Histological evaluation of maxillary sinus mucosa after functional endoscopic sinus surgery. Am J Rhinol 2007;21(6):719-24. [ Özet ]
27) Chan KH, Abzug MJ, Coffinet L, Simoes EA, Cool C, Liu AH Chronic rhinosinusitis in young children differs from adults: a histopathologic study. J Pediatr 2004;144(2):206-12. [ Özet ]
28) Chan Y, Kuhn FA An update on the classifications, diagnosis, and treatment of rhinosinusitis. Current Opin Otolaryngol Head Neck Surg 2009;17(3):204-8. [ Özet ]
29) Ishitoya J, Sakuma S, Tsukuda M Eosinophilic Chronic Rhinosinusitis in Japan. Allergol Int 2010;59(3):239-45. [ Özet ]
30) Schubert MS Allergic fungal sinusitis: pathophysiology, diagnosis and management. Med Mycol 2009;47 Suppl 1:S324-30. [ Özet ]
31) Glass D, Amedee RG Allergic fungal rhinosinusitis: a review. Ochsner J 2011;11(3):271-5. [ Özet ]
32) Ferguson BJ Categorization of eosinophilic chronic sinusitis. Curr Opin Otolaryngol Head Neck Surg 2004;12(3):237-42. [ Özet ]
33) Yoon JH, Na DG, Byun HS, Koh YH, Chung SK, Dong HJ Calcification in chronic maxillary sinusitis: comparison of CT findings with histopathologic results. AJNR Am J Neuroradiol 1999;20(4): 571-4. [ Özet ]
34) Callegas CA, Douglas RG Fungal rhinosinusitis: what every allergist should know. Clin Exp Allergy 2013;43(8):835-49. [ Özet ]
35) Singh V Fungal rhinosinusitis: Unravelling disease spectrum. J Maxillofac Oral Surg 2019;18(2):164-79. [ Özet ]
36) Ho CF, Lee TJ, Wu PW, Huang CC, Chang PH, Huang YL, Lee YL Huang CC Diagnosis of maxillary sinus fungus ball without intralesional hyperintensity on computed tomography. Laryngoscope 2019;129(5):1041-45. [ Özet ]
37) Paparella M, Schachern P, Cureoglu S Chronic silent otitis media. ORL J Otolaryngol Relat Spect 2002;64(2):65-72. [ Özet ]




