SUBMANDIBULAR GLAND INVOLVEMENT IN PATIENTS WHO UNDERWENT LEVEL 1b NECK DISSECTION FOR ORAL CAVITY CANCERS
2Samsun Eğitim ve Araştırma Hastanesi, Patoloji Kliniği, Samsun, Turkey
Summary
Objectives: We aimed to investigate the involvement of submandibular glands (SMGs) in patients who underwent level 1b neck dissection for oral cavity cancer (OCC).Material and Methods: The records of OCCs in which we performed elective level 1b neck dissection between 2014 and 2018 were retrospectively reviewed. Patients who underwent revision or complementary surgery or salvage surgery after radiotherapy, who had concomitant secondary primary or synchronous cancers or diagnosis other than squamous cell carcinoma, patients with unknown tumor size, status of SMG and metastasis level in neck were excluded. A total of 44 patients (n: 44) were included in the study. Primary tumor sub-region rates, neck dissection type, neck metastasis rates, metastasis level, excised SMG number and their involvement and tumor stage were investigated.
Results: The mean age of the patients was 64.55 ± 12.053 (range: 32-86) years and 25.0% (n: 11) of the patients were female and 75.0% (n: 33) were male. The most commonly performed neck dissection (75%) was supraomohyoid (9 bilateral and 22 unilateral). Neck metastasis was seen in 11.4% of the patients (n: 5), and all were in the same side at level 1b region. Among those 2 (40%) were located in lower lip, 2 (40%) were in buccal mucosa and 1 (20%) was on tongue. A total of 61 SMGs were examined in 44 patients and involvement was not observed in any of them.
Conclusion: SMG involvement in level 1b dissections performed for OCC treatment is absent or minimal. In view of the increasing importance of organ sparing surgeries in the current cancer treatment, we think that more comprehensive studies are warranted to investigate the sustainability of SMGs.
Introduction
Oral cavity cancer (OCC) is one of the most common cancers worldwide, especially in developing countries. Squamous cell carcinoma is the most common histological type and the most commonly accused etiological factors are alcohol and tobacco use. [1] The oral cavity (OC) is anatomically consisted of some sub-regions very close to each other including lip, tongue (2/3 anterior), the floor of the mouth (FOM), buccal mucosa, lower and upper gingiva, retromolar trigone and hard palate and each of these sub-regions has different characteristics to be considered for oncological aspects.OC is an important component of the upper respiratory and digestive system, and multidisciplinary treatment regimens should be developed individually in order to minimize the disruption of functions and provide the oncological control in treatment of cancers of this region. The widespread and up-to-date treatment for OCC is the appropriate neck dissection based on the extensive excision of the primary tumor and the extent of neck involvement. [2]
Over the years, neck dissections have become more functional and conservative with similar regional control rates. [3,4] Although it is known that preservation of non-lymphoid structures such as vessels, nerves and muscles does not impair oncologic safety, SMG is almost routinely removed during level 1b dissections due to its proximity to the surrounding lymphoid structures and primary tumor. [2,3] Unlike the parotid gland, SMG does not contain lymph nodes and its involvement in OCC, especially in the early stages, is extremely rare. Therefore, since SMG involvement is expected only with direct invasion, it is said that SMG can be preserved in cases where direct invasion is not seen. [4] First-line lymphatic structures in OCC metastases are at level 1b, so it is important to clear all lymphatics to prevent neck recurrence. Already in level 1 b dissection, the reason for routine removal of SMG is not its involvement with the tumor, but by this way to be able to clean periglandular, especially perivascular lymph nodes, without leaving them behind. Dissection by leaving SMG in place can sometimes be difficult and requires surgical experience and skill. Risking oncological safety in order to protect SMG is of course not the right approach, but preserved SMG will have very important tasks, especially in the case of functionally impaired OC at postoperative period.
SMG is responsible for 70-90% of un-stimulated saliva, especially at nights. [2] Saliva is not only for the wetting of the OC; it is also required for antimicrobial activity in the mouth, re-mineralization of the teeth, maintenance of oral mucosal immunity, and preparation of the bolus during chewing. If salivation is reduced, problems such as dry mouth (xerostomia), mucositis, gingival diseases and tooth decays, cracks in the tongue, taste disorder, discordance of dentures and difficulties in chewing and swallowing may occur. [5] Jaguar et al. [6] have shown in their studies that extracted SMG causes decrease in un-stimulated salivary secretion resulting in xerostomia, and contralateral SMG cannot compensate for it.
In this study, we investigated the involvement of SMG in patients who underwent level 1b dissection for OCC. In our series, lip cancers were more common than other sub-regions and in this regard, we think that we will make a different contribution to the literature.
Methods
After obtaining ethical approval (TUEK 95-2018 BADE / 9-54) of the Medical Education Board Decisions of our Institution, oral cavity tumors that we applied level 1b neck dissection between 2014 and 2018 were retrospectively screened with the relevant surgical codes over digital database.Histopathological reports of our patients with squamous cell oral cavity cancer who underwent uni- or bilateral suprahyoid / supraomohyoid level 1b neck dissection including SMG as the primary treatment were re-investigated. The staging of tumors was performed according to the 8th version TNM staging of the American Cancer Committee. The records of patients who only underwent primary tumor excision, or who had revision or subsequent complementary surgeries were excluded. Patients who underwent salvage surgery after radiotherapy, who had concomitant primary or synchronous cancers, who had a histopathological diagnosis other than squamous cell cancer, and whose histopathology reports did not indicate tumor size, SMG status and neck metastasis level were excluded. Histopathological evaluation was re-performed by the same pathologist as a single blind and correlated with recorded pathology reports. Primary tumor sub-region rates, neck dissection type, neck metastasis rates, metastasis level, number of excised SMG and their involvement and tumor stage were investigated.
Results
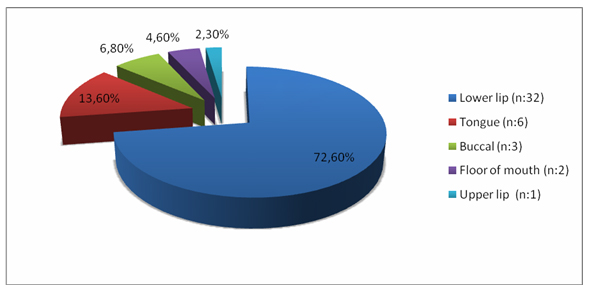
A total of 44 patients (n: 44) who met the inclusion criteria were included in the study. The mean age of the patients was 64.55 ± 12.053 (range: 32-86) years and 25.0% (n: 11) of the patients were female. The most common neck dissection type (75%) was supraomohyoid (9 bilateral, 22 unilateral). Of the 44 patients, 27 (61%) had unilateral dissection and 17 (39%) had bilateral level 1b dissection, so a total of 61 submandibular glands were examined.In our series, the most common primary sub-region was the lower lip (n: 32, 72.7%) and followed by tongue (n: 6, 13.6%), buccal (n: 3, 6.8%), the FOM (n: 2, 4.6%) and upper lip (n: 1, 2.3%). In total 44 (n: 44) patient met the inclusion criteria. (Figure 1)
 Büyütmek İçin Tıklayın |
Figure 1: Distribution of primary tumor localization rates in OCC |
Neck metastasis was seen in 11.4% (n: 5) of the patients, and all were in the same side at level 1b region and none of them had extracapsular spread. Two (40%) tumors of the neck metastasis were located in the lower lip (T2N2bM0), 2 (40%) were on the buccal mucosa (T2N1M0) and 1 (20%) was located on the tongue (T2N2bM0) (Table 2). The tumors with the highest risk of metastasis were found to be buccal mucosa (2/3, 66.6%), tongue (1/6, 16.6%) and lower lip (2/32, 6.2%) tumors, respectively. A total of 61 submandibular glands were examined in 44 patients and involvement was not observed in any of them. (Table 1) The majority of our series (n: 37, 85%) consisted of early stage (stage 1-2) tumors. There were four stage 3 tumors (2 (4.6%) lower lip (T3N0M0) and 2 (4.6%) buccal mucosa) (T2N1M0) in total, and 2 (50%) had metastasis to the neck. In our series, there were three Stage 4 tumors, 2 (4.6%) lower lip (T2N2bM0) and 1 (2.2%) tongue (T2N2bM0). (Table 2)
Table 1: Evaluation of neck metastasis and SMG involvement according to primary regions
Table 2: Classification of primary tumors according to TNM 8
Discussion
SMG is usually removed with lymphatic structures in neck dissections for OCC because of its proximity to the lymphatic structures and primary tumor at level 1b. It is widely accepted that this approach facilitates neck dissection. [3] SMG is located in an aponeurotic sheath consisting of deep and superficial layers. The lymphatic system develops embryologically after the closure of SMG with this sheath. Therefore, unlike the parotid gland, SMG and sublingual gland do not contain lymph nodes and lymphatic ducts in the parenchyma. [7] Although Rouviere [8] identified 5 groups of lymph nodes in this region (preglandular, prevascular, retrovascular, retroglandular and intraglandular), they had a suspicious approach to intraglandular lymph nodes. DiNardo [9] also identified 6th group (deep submandibular) lymph nodes, but in 10 cadaver dissections, he was unable to show lymph nodes within the SMG. Dhiwaker et al. [10] found no lymph nodes either inside or deeper in SMG. Therefore, intraglandular and deep group nodes have little clinical significance because they are absent or very rarely present in some cases. [5] The most common metastatic group is suggested to be the perivascular (prevascular, retrovascular) group. [9]Although not widely accepted, there have been studies suggesting auto-transplantation and subsequent re-implantation of SMG to protect against possible harm to radiotherapy. [5,11] Although sacrificing SMG because of the possibility that it will be harmed with postoperative adjuvant radiotherapy is not suitable for surgical principles, nowadays, radiotherapies can be applied with less harm to SMGs when required by intensity-regulated radiotherapy (IMRT) technology; and this development has further enhanced the importance of the protection of SMG. [3.12] Malik et al. [13] detected level 1b metastasis in 30 of 137 patients with OCC in their prospective study and showed that they had metastasis to deep lymph node group in 3 of them, 2 of which were in the lower gingiva and 1 was in the buccal mucosa. In this series, the majority of which were early-stage tongue tumors, none of the tongue tumors had metastasis to the deep lymph node group, and it was concluded that SMG does not need to be removed in order to dissect deep lymph nodes in OCC, especially in early stage tongue tumors. Metastasis to SMG from both outside the head and neck region by hematogenous pathway and from primary head and neck region were found to be almost non-existent. [2,5,11,14] Vessecchia et al. [15] in a literature review showed that in more than 100 cases metastasizing to SMG, most of them were from distant primers such as breast, lung and genitourinary system by hematogenous way. Basaran et al. [12] found that SMG involvement was seen in 0.6-4.5% of OCC, and as in their series, the most common mechanism was direct invasion and a second mechanism was by metastatic lymph nodes. The third and most commonly discussed mechanism is SMG involvement through intraglandular lymph nodes metastasis, which could not go beyond a few cases. [2,12,16] Because of their anatomic proximity, through direct invasion, the most common tumors of the FOM and tongue have been shown to involve SMG, most of which are advanced (T3-T4) tumors. [2,5,11,12,14] Metastatic lymph nodes have been shown to involve SMG very rarely, although level 1 b metastasis is common in OCC. [2,3,5,11,12,14] It has been shown in pathology studies that the metastatic disease cannot progress to the parenchyma due to the effective barrier effect of the surrounding fibrous capsule despite compressing the gland by pushing. In addition, in a study on 9 metastatic diseases with the main vessels, muscle and skin involvement and macroscopic extracapsular spread, SMG parenchyma was reported not to be involved. [17]
Junquera et al. [16] found the metastasis rate on the same side as 31.7% in 31 patients with oral cavity cancer, but SMG involvement was not detected in any of them. Guney and Yigitbasi [7] concluded that preservation of SMG as a result of functional suprahyoid neck dissection in early stage lip cancer patients does not impair oncologic safety.
In our series, 11.4% of the patients had neck metastasis (n: 5), all were on the same side at the level 1b region, and none had extracapsular spread. Two (40%) of the tumors metastasing to the neck were located in the lower lip (T2N1M0, T3N1M0), 2 (40%) were located in the buccal mucosa (T2N1M0, T2N1M0) and 1 (20%) was on the tongue (T1N1M0). The risk of metastasis to level 1 b was buccal mucosa (2/3, 66.6%), tongue (1/6, 16.6%) and lower lip (2/32, 6.2%), respectively. Fives et al. [18] reported the level 1b involvement rates, respectively, for buccal mucosa (4/5, 80%), lower lip (3/4, 75%), the FOM (13/21, 62%), lower gingiva (4/7, 57%), and tongue (4/21, 19%). Although our rates for buccal mucosa and tongue overlap, the number of lip tumors in our series seems to be high and most of them being early stage with lower metastasis rates. Fives et al. [18] in that study, did not find any lymph nodes in any of the SMGs, but reported perhaps a fourth mechanism to consider for SMG involvement, namely; in a tumor located primarily in the midline of the FOM although pathological involvement of both neck lymph nodes could not be demonstrated bilateral SMG involvement was observed and detected in situ carcinoma in the Wharton canal close to both involved gland stromas and hypothesized that the tumor had spread to the SMG via bilateral channels.Naidu et al.[19] described SMG involvement in only two patients in their series of mostly advanced FOM and tongue cancers and concluded that SMG could be preserved in early stage tumors.. Ashfaq et al. [20] showed that only 2 SMGs were invaded by direct invasion in early stage OCCs, most of which consisted of tongue and FOM cancers. Panda et al. [21] examined a total of 163 SMGs in their series consisting mainly of the tongue, buccal mucosa and gingival sub-regions, and showed that 4 had direct invasion, 1 had level 1b metastatic lymph nodes by extracapsular invasion and 1 had involvement by both mechanisms. They concluded that SMG can be preserved in cases where significant involvement of SMG clinically, radiologically or intraoperatively is not seen and this does not affect survival. Subramaniam et al. [22] described the> 4 cm tumor size and> 10 mm depth of invasion as a risk factor in direct invasion of SMG, and stated that buccal mucosa tumors were 3 times more risky and FOM tumors were 4 times more risky than tongue tumors, due to anatomical proximity. For that reason, they argued that the sub-region is an important marker. Cetin et al. [23] suggested that SMG can be preserved in early stage OCC unless the primary tumor originates or spreads from the FOM.
In each of the past studies with OCC, different sub-regions were evaluated at different rates or at different stages, and lip cancers were either little or almost not involved (Table 3). Since lip cancers have a different historical background, they were separated from OCC for a period of time. [24] Or this may be because it is not as common as in our country. Although lip cancers have been perceived as a skin cancer due to different risk factors such as prolonged exposure to sunlight and having fair skin, they generally show a clinical and pathological characteristic between less aggressive skin cancer and more aggressive oral cavity cancers. [25,26] In its 7th edition published in 2010, the Cancer Staging Manual of the American Joint Committee on Cancer (AJCC) classified lip cancers as a special subtype of oral cavity cancers. [27]
The majority of our series (n: 37, 85%) consisted of early stage (stage 1-2) tumors. Unlike other series, early stage lower lip tumors (n: 28, 63.6%) were the majority of them. The highest number of lip cancers as the primary sub-region (n: 32) was present in our series (Table 3), and totally 61 submandibular gland examinations revealed no involvement.
SMG, due to the salivation it largely assumes (70-90%); provides wetting of the oral cavity and also makes a significant contribution to oral antimicrobial activity, remineralization of teeth, maintenance of oral mucosal immunity, and bolus preparation during chewing. [5,6]
It should be kept in mind that the preservation of SMG will also have advantages such as avoiding marginal mandibular nerve (7.7%), hypoglossal nerve (2.9%) and lingual nerve (1.4%) damage during the surgery of the gland and avoiding cosmetic complaints due to the gaps in the removed gland. [13]
In conclusion, it should be kept in mind that performing level 1b dissection while preserving SMG is a procedure that requires surgical experience and skill, although there is sufficient reason for the preservation of SMG. Even though it is ideal to maintain SMG without risking oncologic safety, if the lymphatic structures in level 1b cannot be cleared without removal of SMG, SMG subtraction would be a more appropriate approach for oncologic safety.
Conclusion
Especially in early stage lip cancers, as in other oral cavity tumors, SMG involvement is very rare. Given the growing importance of organ sparing surgeries in today's cancer treatment, we think that more attention should be paid to the preservation of SMG and for this purpose prospective studies are needed to investigate the oncological safety of SMG protection.
Declaration of conflicting interests:
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding:
The authors received no financial support for the research and/or authorship of this article.
Reference
1) Montero PH, Patel SG. Cancer of the oral cavity. Surg Oncol Clin N Am. 2015; 24:491-508. [ Özet ]
2) Chen TC, Lo WC, Ko JY, Lou PJ, Yang TL, Wang CP. Rare involvement of submandibular gland by oral squamous cell carcinoma. Head Neck. 2009; 31:877-81. [ Özet ]
3) Takes RP, Robbins KT, Woolgar JA, Rinaldo A, Silver CE, Olofsson J, Ferlito A. Questionable necessity to remove the submandibular gland in neck dissection. Head Neck. 2011; 33:743-5. [ Özet ]
4) Coskun HH, Medina JE, Robbins KT, Silver CE, Strojan P, Teymoortash A, Pellitteri PK, Rodrigo JP, Stoeckli SJ, Shaha AR, Suárez C, Hartl DM, de Bree R, Takes RP, Hamoir M, Pitman KT, Rinaldo A, Ferlito A. Current philosophy in the surgical management of neck metastases for head and neck squamous cell carcinoma. Head Neck. 2015; 37:915-26. [ Özet ]
5) Byeon HK, Lim YC, Koo BS, Choi EC. Metastasis to the submandibular gland in oral cavity squamous cell carcinomas: pathologic analysis. Acta Otolaryngol. 2009; 129:96-100. [ Özet ]
6) Jaguar GC, Lima EN, Kowalski LP, Pellizon AC, Carvalho AL, Alves FA. Impact of submandibular gland excision on salivary gland function in head and neck cancer patients. Oral Oncol. 2010; 46:349-54. [ Özet ]
7) Guney E, Yigitbasi OG. Functional surgical approach to the level I for staging early carcinoma of the lower lip. Otolaryngol Head Neck Surg. 2004; 131:503-8. [ Özet ]
8) Rouviere H, Tobies MJ. Trans: Anatomy of the human lymphatic system. Ann Arbor, MI: Edwards Brothers. 1938; p. 86-8.
9) DiNardo LJ. Lymphatics of the submandibular space: an anatomic, clinical, and pathologic study with applications to floor-of-mouth carcinoma. Laryngoscope. 1998; 108:206-14. [ Özet ]
10) Dhiwakar M, Ronen O, Malone J, Rao K, Bell S, Phillips R, Shevlin B, Robbins KT. Feasibility of submandibular gland preservation in neck dissection: a prospective anatomicpathologic study. Head Neck. 2011; 33:603-609. [ Özet ]
11) Spiegel JH, Brys AK, Bhakti A, Singer MI. Metastasis to the submandibular gland in head and neck carcinomas. Head Neck. 2004; 26:1064-8. [ Özet ]
12) Basaran B, Ulusan M, Orhan KS, Gunes S, Suoglu Y. Is it necessary to remove submandibular glands in squamous cell carcinomas of the oral cavity? Acta Otorhinolaryngol Ital. 2013; 33:88-92. [ Özet ]
13) Malik A, Joshi P, Mishra A, Garg A, Mair M, Chakrabarti S, Nair S, Nair D, Chaturvedi P. Prospective study of the pattern of lymphatic metastasis in relation to the submandibular gland in patients with carcinoma of the oral cavity. Head Neck. 2016; 38:1703-1707. [ Özet ]
14) Razfar A, Walvekar RR, Melkane A, Johnson JT, Myers EN. Incidence and patterns of regional metastasis in early oral squamous cell cancers: feasibility of submandibular gland preservation. Head Neck. 2009; 31:1619-23. [ Özet ]
15) Vessecchia G, Di Palma S, Giardini R. Submandibular gland metastases of breast carcinoma; a case report and review of the literature. Virchows Arch.1995; 427:349-51. [ Özet ]
16) Junquera L, Albertos JM, Ascani G, et al. Involvement of the submandibular region in epidermoid carcinoma of the mouth floor. Prospective study of 31 cases. Minerva Stomatol. 2000; 49:521-5. [ Özet ]
17) Woolgar JA, Triantafyllou A. Pitfalls and procedures in the histopathological diagnosis of oral and oropharyngeal squamous cell carcinoma and a review of the role of pathology in prognosis. Oral Oncol. 2009; 45:361-385. [ Özet ]
18) Fives C, Feeley L, Sadadcharam M, O'Leary G, Sheahan P. Incidence of intraglandular lymph nodes within submandibular gland, and involvement by floor of mouth cancer. Eur Arch Otorhinolaryngol. 2017; 274:461-466. [ Özet ]
19) Naidu TK, Naidoo SK, Ramdial PK. Oral cavity squamous cell carcinoma metastasis to the submandibular gland. J Laryngol Otol. 2012; 126:279-84. [ Özet ]
20) Ashfaq K, Ashfaq M, Ahmed A, Khan M, Azhar M. Submandibular gland involvement in early stage oral cavity carcinomas: can the gland be left behind? J Coll Physicians Surg Pak. 2014; 24:565-8. [ Özet ]
21) Panda NK, Patro SK, Bakshi J, Verma RK, Das A, Chatterjee D. Metastasis to submandibular glands in oral cavity cancers: Can we preserve the gland safely? Auris Nasus Larynx. 2015; 42:322-5. [ Özet ]
22) Subramaniam N, Balasubramanian D, Reddy R, Rathod P, Murthy S, Vidhyadharan S, Thankappan K, Iyer S. Determinants of level Ib involvement in oral squamous cell carcinoma and implications for submandibular gland-sparing neck dissection. Int J Oral Maxillofac Surg. 2018; 47:1507-1510. [ Özet ]
23) Cakir Cetin A, Dogan E, Ozay H, Kumus O, Erdag TK, Karabay N, Sarioglu S, Ikiz AO. Submandibular gland invasion and feasibility of gland-sparing neck dissection in oral cavity carcinoma. J Laryngol Otol. 2018; 132:446-451. [ Özet ]
24) Kerawala C, Roques T, Jeannon JP, Bisase B. Oral cavity and lip cancer: United Kingdom National Multidisciplinary Guidelines. J Laryngol Otol. 2016; 130:83-S89. [ Özet ]
25) Biasoli ÉR, Valente VB, Mantovan B, Collado FU, Neto SC, Sundefeld ML, Miyahara GI, Bernabé DG. Lip Cancer: A Clinicopathological Study and Treatment Outcomes in a 25-Year Experience. J Oral Maxillofac Surg. 2016; 74:1360-7. [ Özet ]
26) Tseng HW, Liou HH, Tsai KW, Ger LP, Shiue YL. Clinicopathological study of lip cancer: a retrospective hospital-based study in Taiwan .APMIS. 2017; 125:1007-1016. [ Özet ]
27) Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17:1471-4. [ Özet ]




