HISTOPATHOLOGIC EVALUATION OF THE EFFECTS OF SYSTEMIC METHYL-PREDNISOLONE TREATMENT ON EOSINOPHILIC NASAL POLYPOSIS
Summary
Aim: To study the histopathologic effects of systemic cortisone treatment on eosinophilic nasal polyposis.Material and method: Systemic corticosteroid treatments were used in 25 patients with nasal polyposis. An oral dose of 1mg/kg prednisolone was given for the first 5 days, than the steroid was tapered in 17 days by decreasing it's dose 5mg per day Nasal mucosal biopsies taken before and after the treatment were evaluated histopathologically.
Results: Following medical treatment, a significant reduction in edema and eosinophilia, and an increase in neutrophilia were detected (p<0.05). Other histopathological parameters showed no significant change (p>0.05).
Conclusion: Corticosteroid treatments have limited therapeutic effect by histopathologic level on nasal polyposis.
Introduction
Nasal polyposis (NPs) is a chronic inflammatory disease of the paranasal sinuses, which primarily arises from the anterior ethmoid cells, and causes symptoms such as congestion, rhinorrhea, hyposmia and a feeling of fullness in the face. The histopathologic features of NPs are characterized by dense edematous extracellular connective tissue and subepithelial regional mast cells, with inflammatory cell infiltration comprised of eosinophil, lymphocyte, neutrophil and plasma cells[1]. The superficial epithelium is located on an almost indistinguishable basement membrane. There is a thick basement membrane, an increased number of goblet cells and scattered submucosal glands. In general, eosinophils dominate in 80% of polyps, while neutrophils become predominant in cases with infection[2,3]. Tissue eosinophilia is a hallmark of nasal polyposis however there is no universally accepted definition for eosinophilic nasal polyp[4-5]Medical treatment of NPs mostly includes topical and systemic corticosteroids. Steroids bind to specific glucocorticoid receptors, and exert anti-inflammatory effects by decreasing the number, lifespan and activity of the inflammatory cells like eosinophils, mast cells, basophiles, and T and B lymphocytes. Although systemic steroids are generally considered effective in NP treatment, there are only a few researches in this issue, and these studies are usually based on radiological or endoscopic observations. In this study, our aim was to assess effects of corticosteroid treatment on NPs histopathologically.
Methods
The study was carried out in NPs patients who admitted to the Ear, Nose and Throat Clinic of Ataturk Training and Research Hospital between 2009 - 2010. The patients included in the study had Grades 2 and 3 NPs according to the Mackay and Naclerio classification. They had no contraindication to systemic corticosteroid treatment (diabetes mellitus, gastritis, gastric ulcer, cardiac disease, generalized osteoporosis, etc), and did not have any previous surgery for NPs. Written informed patient consents from all patients were obtained and local ethical committee approved the study. Patients with cystic fibrosis, ciliary dyskinesia, asthma, nasal polyposis, antrochoanal polyps and unilateral NPs were not included in the study No radiological evaluation was performed prior to systemic corticosteroid treatment.Prior to treatment, polyp samples were obtained from all patients under local anesthesia for histopathologic evaluation. These biopsies comprised of Group 1. In the treatment, each patient was given an oral dose of 1mg/kg prednisolone for the first 5 days, than the steroid was tapered in 17 days by decreasing its dose 5mg per day.
One week after completion of the treatment, coronal plane paranasal sinus computerized tomography (PNCT) was obtained. The patients who did not have recovery on PNCT as well as relief of symptoms like nasal obstruction, postnasal dripping were treated with functional endoscopic sinus surgery (FESS). There were 25 patients who had FESS. During surgery, biopsies were obtained from polyp samples for histopathologic evaluation. These biopsies comprised of Group 2.
Histopathology: On histopathology, the polyp samples were stained with Hematoxylin-Eosin (H-E) and inflammatory parameters like edema, basal membrane thickness, neutrophil, eosinophil, lymphocyte, plasma cell, and goblet cell changes, seromucinous gland hyperplasia and epithelial erosion were investigated. All specimens were evaluated by the same pathologist.
Under light microscope at x400 magnification, mostly dense stained area was selected by the pathologist who was blinded to the sample, and the cells were counted. When the number of eosinophil cells exceeded 20% of all cells counted (eosinophils and inflammatory cells), this condition was considered positivity. Otherwise the result of the evaluation was considered negativity. The samples were also investigated for the presence of edema, plasma cells, basal membrane thickening, seromucinous gland hyperplasia and epithelial erosion. Basement membrane thickening was measured and recorded as positive for thickening over 10 µm and negative for thinner than 10 µm. The presence of plasma cells, edema, seromucinous gland hyperplasia and epithelial erosion were evaluated as positive or negative lymphocytes and neutrophils were considered positive for the groups create the most intense cell. Pre- and post treatment findings were compared statistically (Table-I).
Table 1: Histopathologic findings of the patients
Statistical Analyses: Data analysis was performed by using SPSS for Windows, version 11.5. Nominal data were expressed as number of patients and percentages. Pre and post treatment data were compared using Mc Nemar test. A p value less than 0.05 was considered statistically significant.
Results
Of 25 patients, 17 were males and 8 were females, with a mean age of 40.1-+15.5 years. No serious side effect of systemic steroid was seen. In the surgical treatment, 11 patients were treated with anterior ethmoidectomy, 9 with total ethmoidectomy and frontal recess surgery and 5 with total ethmoidosphenoidectomy. No major complication occurred in the surgeries. Histopathologic findings of both Groups are shown in Table-1.On histopathologic investigation, edema was positive in 24 of 25 patients (96%), negative in 1 patient (4%) in Group 1 and was positive in 7 of 25 patients (28%) and negative in 18 patients (72%) in Group 2. A statistically significant difference was found between the groups (P= 0.031) (Figure 1-2).
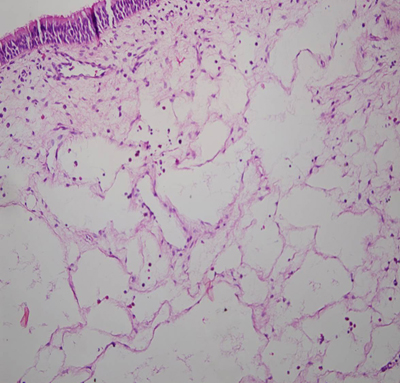 Büyütmek İçin Tıklayın |
Figure 1: Histopathologic view of a polyp with edema and eosinophilia |
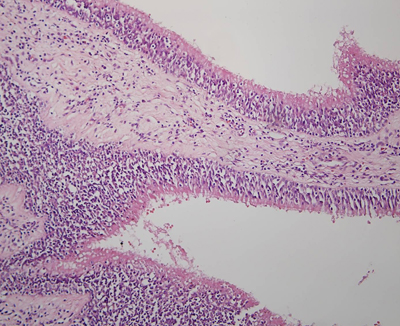 Büyütmek İçin Tıklayın |
Figure 2: Histopathologic view of a polyp without edema with eosinophilia |
The predominance of the eosinophils among the inflammatory cells was evaluated. In Group-1, eosinophil predominance was seen in 23 patients (92%) while there was negativity in 2 (8%) of the patients. In Group-2, 9 (36%) of 25 patients did not have eosinophil predominance, while in remaining 16 (64%) patients the predominance continued. The difference between the groups was statistically significant (P=0.016). The eosinophil counts were found significantly dropped statistically after steroid treatment.
As for the neutrophils; in group-1, 11 (44%) of the patients were positive and 14 (56%) were negative. In Group-2, 2 (8%) of the patients were negative and 23 (92%) were positive. The difference between the groups was statistically significant (P=0.004).None of the remaining parameters were significantly different between the two groups (Table-1).
Discussion
Etiopathology of NPs is unknown. However, there is an evolution in the medical and surgical treatment modalities of NP[6,7]. Interleukin-4 (IL.4) increase due to viral, bacterial or allergic factors, and some other mediators may cause proliferation of submucosal stroma, thereby increased vascular permeability may cause the edematous stroma seen in NP. IL-6, IL-8 and IL-11 are indicators of nonspecific inflammation[8]. In addition, eosinophils can potentiate the inflammatory process by causing plasma exudation due to increased vascular permeability. That increase in the inflammatory mediators can lead to development of NPs. Tissue edema resulting from fluid leakage out of the vessels plays an important role in the development of NPs. According to Soler ZM et al. mucosal eosinophilia correlates with objective disease severity as defined by CT, endoscopy and smell identification test.[9]Watanabe et al[10] investigated anti-inflammatory effects of topical steroids on NPs both in vivo and in vitro. Accordingly, they reported that the clinical efficacy of glucocorticoids in NPs may be due to induction of apoptosis of local eosinophils and neutrophils. This apoptosis may be a direct effect, or may occur indirectly due to down-regulation of granulocyte-macrophage colony-stimulating factor (GM-CSF) production in the epithelial cells of NPs (NPECs). A similar mechanism also functions in systemic steroid treatment[11]. In more than half of the NP patients, the steroid treatment decreases the extent of polyposis[12]. Steroids cause a symptomatic relief in NP, which also become evident on the CT scans of the paranasal sinuses[13].
According to Hellquist et al[14], histopathological diagnosis of NP can be made in the presence of eosinophilia, chronic inflammatory process, seromucinous gland atrophy and atypical stromal polyps. According to Mygind et al[3], nasal eosinophilia and steroid responsiveness are the features of eosinophilic polyps while neutrophil polyps, which suggest an infectious source, are characterized by the presence of neutrophils, purulent secretion and steroid resistance. Histopathological patterns of the eosinophilic polyps are stromal edema, goblet cell hyperplasia, dense eosinophilia and mast cells accumulation in the stroma. In addition, there is a thick hyalinised basal membrane between the epithelium and stroma.
Currently there is no definite histological criterion for differentiation of eosinophilic nasal polyp[5] Jeong WJ[4] et al. suggested a tissue eosinophil proportion of more than 11 % as a criterion for eosinophilic allergic polyp. In this research our criteria for eosinophilic polyp was 20 %.
Van Camp and Clement[13] observed a symptomatic relief with a high dose prednisolone in 25 NP patients, which was also evident in half of the patients on CT scan. The authors proposed that systemic steroid treatment must be performed for surgical candidates and preoperative systemic steroid treatment facilitates surgical intervention. According to Takeno S[15] et al. the objectives of eosinophilic chronic rhinosinusitis management should focus directly on inhibition of focal eosinophil infiltration. The use of local and/or systemic steroids is a potent treatment strategy for preventing relapse of nasal polyposis and is considered to be the first line treatment for eosinophilic chronic rhinosinusitis patients. These proposals are parallel with our findings. In our study, eosinophilic polyps responded properly to steroid treatment. The patients had symptomatic relief in addition to decreased polyp size on nasal endoscopy. This clinical observation was also supported histopathologically where there was a decrease in tissue edema and number of the eosinophils. However, an increase in the numbers of neutrophils was observed after steroid treatment. This condition may be attributed to an infectious process. This contention is also supported by the increased numbers of lymphocytes and plasma cells which indicate a progressive chronic infectious condition.
In conclusion, corticosteroid treatment seems to have a limited effect on NP at histopathological level despite the presence of clinical relief in the patients.
Reference
1) Larsen K, Tos M. The estimated incidence of symptomatic nasal polyps. Acta Otolaryngol 2002; 122:179-182. [ Özet ]
2) Hamilos DL, Leung DY, Wood R, Meyers A, Stephens JK, Barkans J, Meng Q, et al. Chronic hyperplastic sinusitis: association of tissue eosinophilia with mRNA expression of granulocyte-macrophage colony-stimulating factor and interleukin-3. J Allergy Clin Immunol 1993; 92:39-48. [ Özet ]
3) Mygind N. Nasal polyps. In: Mygind N. Nasal Allergy. London: Blackwell Scientific publiactions1978:233-238.
4) Jeong WJ, Lee CH, Cho SH, Rhee CS. Eosinophilic allergic polyp: a clinically oriented concept of nasal polyp. Otolaryngol Head Neck Surg. 2011 Feb; 144(2):241 -6 [ Özet ]
5) Eghtedari F, Cheraghzadeh SR, Kashef MA, Monabati A, Kashef S. Agreement rate of skin prick test with tissue eosinophil count in patients with nasal polyps. Iran J Allergy Asthma Immunol.2007 Jun;6(2):89-92. [ Özet ]
6) Wigand ME, Hosemann WG. Results of endoscopic surgery of the paranasal sinuses and anterior skull base. J Otolaryngol 1991;20:385-90. [ Özet ]
7) Damm M, Jungehulsing M, Eckel HE, Schmidt M, Theissen P. Effects of systemic steroid treatment in chronic polypoid rhinosinusitis evaluated with magnetic resonance imaging. Otolaryngol Head Neck Surg. 1999; 120(4):517-23. [ Özet ]
8) Bradley DT, Kountakis SE. Role of interleukins and transforming growth factor-beta in chronic rhinosinusitis and nasal polyposis. Laryngoscope. 2005 Apr; 115(4):684-6. [ Özet ]
9) Soler ZM, Sauer DA, Mace J, Smith TL. Relationship between clinical measures and histopathologic findings in chronic rhinosinusitis. Otolaryngol Head Neck Surg.2009 Oct;141(4):456-61. [ Özet ]
10) Watanabe K, Shirasaki H, Kanaizumi E, Himi T. Effects of glucocorticoids on infiltrating cells and epithelial cells of nasal polyps. Ann Otol Rhinol Laryngol. 2004; 113(6):465-73 [ Özet ]
11) Norlander T, Brönnegard M, Stierna P. The relationship of nasal polyps, infection, and inflammation. Am J Rhinol 1999;13(5):349-355. [ Özet ]
12) Drake-lee AB. The value of medical treatment in nasal polyps. Clin Otolaryngol 1991 16(3):237-9 [ Özet ]
13) Van Camp C, Clement PA. Results of oral steroid treatment in nasal polyposis. Rhinology. 1994; 32(1):5-9 [ Özet ]
14) Hellquist HB. Nasal polyps update. Histopathology. Allergy Asthma Proc 1996; 17(5):237-42. [ Özet ]
15) Takeno S, Hirakawa K, Ishino T. Pathological mechanisms and clinical features of eosinophilic chronic rhinosinusitis in the Japanese population. Allergol Int.2010 Sep;59(3):247-56 [ Özet ]




